Mechanosensitivity and Mechanotransduction: 4 (Mechanosensitivity in Cells and Tissues)
Contents:
It is important to note that Collinsworth et al. We developed a method that enabled us to define the transversal stiffness of different parts of the muscle fibre [ 5 ]. Conducted measurements of the fibres of different skeletal muscles and cardiomyocytes show that the transversal stiffness of myocardial fibre membranes, in all parts, are definitively higher than the transversal stiffness of the membrane of skeletal muscle fibres.
The increases in transversal stiffness of the muscle cell membranes are connected to the development of the cytoskeleton, in particular at cell maturation. Evidently, a more developed cortical cytoskeleton can explain the increase in cardiomyocyte stiffness, while with the fibres of skeletal muscles this increase is likely explained by high mechanical load. A similar approach was used by Martens and Radmacher [ 10 ] with the aim of demonstrating the main contributions of the actin cytoskeleton in the stiffness of human fibroblasts, which was assessed before and after addition of blebbistatin and the Rho-kinase inhibitor Y Normal human lymphocytes and human T-lymphoblastic Jurkat cells were examined.
Atomic force microscopic images showed that the cell profiles, that is, surface striations, are similar in both types. Except those mentioned above, the most widely studied cells are those of the human bone marrow, which are represented in particular by mesenchymal stem cells hMSCs and osteoblasts hOBs. Moreover RS and MG63 cells were flatter on the fibrous substrates than on smoothed surfaces on the slide. In contrast, in cells with greater surface area, that is, FC and hOBs, flatness was not dependent on the substrate and was more evident in comparison with RS and MG63 cells.
The authors tried to explain this result as being due to different degrees of cell adhesion, taking into account the higher content of focal adhesion complexes in FC and hOBs than in RS and MG63 cells.
When examining the mechanical properties of these types of cell, Docheva et al. The objects of their study were MC3T3-E1 osteoblast-like cells, tested on the following different substrates: At the time damage of microtubules did not cause any significant effects. A more detailed study of the influence of the extracellular matrix on the morphology and mechanical properties of cells was conducted by Yim et al. From results obtained by Yim et al.
Cell Mechanosensitivity: Mechanical Properties and Interaction with Gravitational Field
Thus, the data of different authors enables us to suggest that the stiffness of nonmuscle cells depends significantly on the type of substrate on which they are cultivated. In addition, the nanotopography of the substrate has a substantial significance that can be connected to the formation of adhesion sites and development of a submembranous cytoskeleton. Data concerning the increased stiffness of proliferated cells, which are also characterised by an increase in protein content forming cortical cytoskeleton, also demonstrates this.
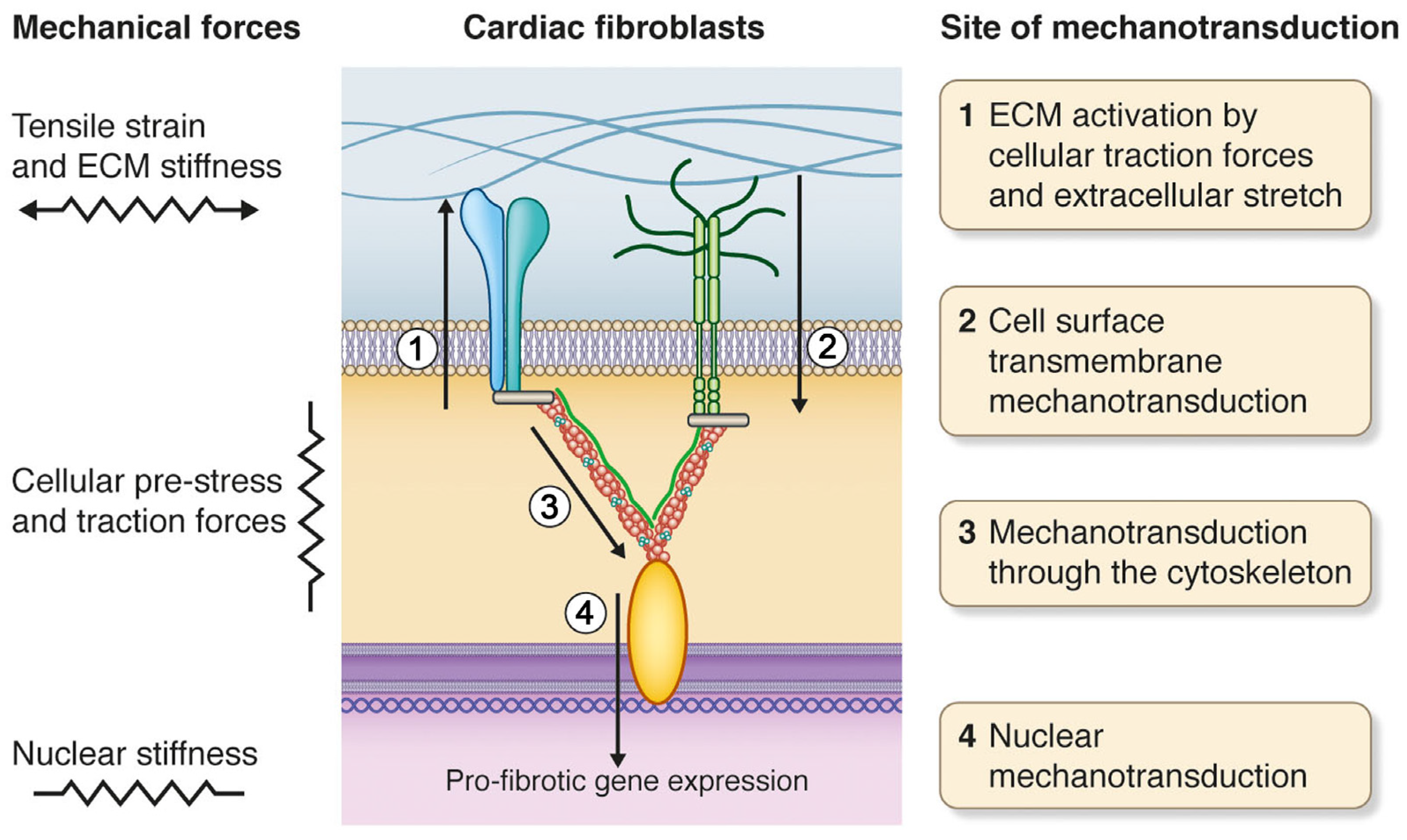
The process of transformation of physical signals into biochemical ones, and the formation of appropriate cellular responses, is called mechanotransduction or mechanosensitivity [ 18 ]. Understanding of the molecular basis of mechanotransduction is impossible without knowledge of the amplitude and distribution of forces influencing cells. However data presented in the literature and conducted in this direction are very limited. First Davies et al. The central idea of these authors is that the force affects more structures, although it becomes lessened with increasing distance from the point of force application.
Nevertheless, this is not always the case as these authors showed in additional work using intermediate filaments as a marker for changing forces [ 20 , 21 ]. Similar experimental approaches were used by several other groups whose purpose was the mapping of stresses that appeared in the cell in response to the applied external force [ 22 — 24 ].
All these results prove that forces have a complex, heterogeneous nature that is mediated by several proteins and their complexes. Force can be exerted on the cell by different experimental methods, and if the applied force is sufficient, the cellular response can be analysed. In this way, Huang et al. It is relatively difficult to compare these values with the force measured in experiments with alternative deformation because there are no measurements of the force of cell interaction and elasticity of the substrate.
The analysis of the force of cell interaction and the substrate without overlapping of information can be the alternative for such measurements. While studying fibroblasts, Balaban et al. Assuming the close packaging of integrins in the focal adhesion complex, the authors assessed the force necessary to cause changes in the conformation of every integrin to be at the level of several pN.
The level of force necessary for a change of conformation in other proteins also can be determined.
The force necessary to break the link between two proteins can be taken as the upper assessment. So for the dissociation of fibronectin from integrin, a force of 30— pN is required [ 26 ]. This is comparable to the magnitude of force generated by one myosin head at muscle contraction [ 29 ], which increases the assurance of the accuracy of the assessments made by Huang et al. It has been shown that the applying of stretching force to a culture of neurons or smooth muscle cells, through the extracellular matrix leads to an increase in the polymerisation of microtubules [ 30 , 31 ].
Integrins, which form connections with various proteins in the extracellular matrix, such as fibronectin and vitronectin, form a primary site of transduction and consequently, can be considered as mechanosensors. These proteins include paxillin, focal adhesion kinase, and caveolin, where paxillin and focal adhesion kinase can connect to a large number of other proteins, thus forming signalling cascades. Besides these, tensin, alpha-actinin, and filamin can also connect with integrins and the cortical cytoskeleton, as they have the appropriate domains, and work in conjunction with integrins and actin [ 32 ].
Furthermore, alpha-actinin has more than one domain with which it interacts with actin and form an actin network. The structure of the focal adhesion complex is characterised by large number of proteins, all located in the immediate vicinity of each other, and so this feature complicates the analysis of the contribution of each in mechanotransduction and does not allow for determinations of which have the more dominant roles.
There was a problem providing the content you requested
However, it is obvious that an external mechanical force can lead to conformational changes in one or several of the proteins of the focal adhesion complex, further triggering the cascade of underlying signalling pathways. Mechanical stretching of cellular membranes, for example using the patch-clamp technique, changes the transportation activity of mechanosensitive ion channels as a result of conformational changes or changes in the lipid bilayer [ 33 , 34 ], or in the gate domains of the channel itself [ 35 , 36 ].
In addition, the majority of channels studied respond to cellular stretching, but not to compression. Prokaryotic mechanosensitive ion channels have been described in many experimental studies and reviews. One of the most well-characterised mechanosensitive channels is the bacterial MscL, which has a pore with a large diameter and low ion selectivity. Increasing the membrane tension, supervised by variation of depth of absorption to a pipette, caused an increase in the conductivity of channels when the forces operating on the channels exceed a certain size [ 37 ].
The results of molecular dynamic modeling [ 38 ], based on the data on MscL crystal structure, show that such changes in membrane tension lead to the formation of pores with a diameter of 0. However, the adequacy of the results of the experimental situations in vivo is under question. Few eukaryotic channels have been identified as mechanosensitive channels: The transient receptor potential TRP protein superfamily consists of a diverse group of cation channels that have important roles in cells of the nervous system and in nonexcitable cells [ 42 ].
It has been shown that the underlying cytoskeleton and scaffolding proteins can influence the regulation of gating in TRP channels [ 43 ]. They are thought to play a major role in setting the resting membrane potential of many cell types. These ion channels are found in different sodium-absorbing epithelia, including the epithelium of the colon, lung, and distal nephron; their activity represents the rate-limiting step for sodium uptake, and thus transepithelial water movement [ 45 ].
There is growing evidence concerning the activation of ENaC by mechanical forces, and at least laminar shear stress seems to be an adequate stimulus of physiological significance [ 46 , 47 ]. Today the role of cortical cytoskeleton in the regulation of ion channels is quite well established. It has been shown that condensation of cortical actin under the plasma membrane occurs as a result of the phosphatase inhibitor, calyculin A, suppressing a depot-dependent input of calcium in smooth muscle cells in culture [ 54 ], and also cytochalasin D [ 55 ].
Dismantling of actin filaments by cytochalasin D leads to the activation of sodium channels in K cell lines, while actin polymerisation on the cytoplasmic side of an external membrane of a cell causes channel inactivation [ 60 ]. Thus, the fragmentation of actin filaments, as associated with a plasma membrane, caused by cytosolic actin-connecting Ca-sensitive proteins that are similar to endogenous gelsolin, can be a major factor in inducing the activity of sodium channels in response to increasing intracellular concentrations of calcium ions in K cell lines [ 61 , 62 ].
In addition to the examples mentioned above, there are data which indicate that an association with the lipid microdomains of cholesterol-rich plasma membranes rafts could be the essential factor in defining the activity of integrated membrane proteins, including ionic channels [ 63 — 68 ]. The disruption of the membrane structure and raft integrity, caused by a decrease in the level of membrane cholesterol, interferes with execution of cellular functions, including reorganisation of the actin network [ 66 , 69 ].
Therefore, it was shown that a partial extraction of membrane cholesterol using methyl-beta cyclodextrin, at concentrations of 2. In cells with a lowered cholesterol content, there was an observed increase in the threshold of activation, and a decrease in the probability of channels being in an open state. Thus, measurements of mechanosensitive flows in various conditions, and complementary data from fluorescent microscopy, indicate that suppression of activity of mechanosensitive channels is mediated by the reorganisation of actin, which is initiated, according to the authors, by disruption of raft integrity due to decreases in the levels of membrane cholesterol [ 70 , 71 ].
Considering that the initiation of many intracellular signalling pathways is a result of membrane-bound proteins that amplify, at increased speed, the lateral diffusion of a signal, Jalali et al. Using fluorescent labels, they estimated the speed of their migration in phospholipids bilayer, upon applying a shearing stress, and the dependence on how this stress was increased, that is, smoothly or in steps. In the case of a step-wise increase, the diffusion factor increased, while in case of gradual increase of stress this decreased.
Cell Mechanosensitivity: Mechanical Properties and Interaction with Gravitational Field
In their review, Huang et al. Nevertheless, it still remains unclear how shearing stress leads to changes in membrane fluidity. It is well known that the action of external forces can lead to changes in the levels of gene expression. In combination of facts that the forces enclosed through the membrane-connected receptors, in certain cases, can lead to nucleus deformations [ 74 ], it is possible to assume a direct influence of external forces on chromatin and, as a result, on expression level [ 18 ].
Forces in this case could be transduced through the cytoskeletal network to the nuclear envelope, and then, through the laminin network to chromatin. In addition, the action of external forces could be transduced to microtubules, leading to their disruption, depolymerisation, and the initiation of a signalling pathway [ 75 ]. It should be noted that conformational changes in various proteins could potentially be applied to a mechanosensory role, but there is no direct, practical proof of this.
However, there is at least one example that a biochemical response can be caused by conformational changes of proteins. As discussed above, the folded domains of fibronectin can be exposed through the application of a stretching force to the molecule, which leads to fibril formation. These levels of force are comparable to that, which according to estimates, can initiate mechanotransduction.
It has been shown that the applying of stretching force to a culture of neurons or smooth muscle cells, through the extracellular matrix leads to an increase in the polymerisation of microtubules [ 30 , 31 ]. Under cell stretch, there are cortical cytoskeleton deformations and subsequent shifts in actin filaments relative to each other in the stress fibres. The change in differentiation potential of stem cells, after real or modelled microgravity changes, can also be connected to cytoskeleton reorganisation, as some of cytoskeletal structures may take part in the determination of the differentiation pathway [ , ]. Thus, the fragmentation of actin filaments, as associated with a plasma membrane, caused by cytosolic actin-connecting Ca-sensitive proteins that are similar to endogenous gelsolin, can be a major factor in inducing the activity of sodium channels in response to increasing intracellular concentrations of calcium ions in K cell lines [ 61 , 62 ]. According to theories proposed by Ingber [ 83 ], the cytoskeleton as a whole reacts to changes in mechanical tension through the extracellular matrix, and the associated integrins, leading to the reorganisation of microfilaments and microtubules. A similar approach was used by Martens and Radmacher [ 10 ] with the aim of demonstrating the main contributions of the actin cytoskeleton in the stiffness of human fibroblasts, which was assessed before and after addition of blebbistatin and the Rho-kinase inhibitor Y
Much less is known, however, concerning the various intracellular proteins e. In fact, any protein participating in a mechanotransduction from extracellular contacts of a cell can act as a mechanosensor and stimulate the unfolding of both integrin isoforms [ 80 ] in addition to the proteins associated with them [ 81 ]. Proteins of the focal adhesion complex are also primary candidates for roles as mechanosensors.
This becomes particularly obvious in the case of experimental data which shows that the stretching of cells, through the use of detergent for removal of the cellular membrane , on a pliable substratum can lead to communication strengthening between focal adhesion kinase and paxillin, in the region of focal adhesion [ 82 ].
As the cellular membrane was removed in these experiments, the ion channels could not participate in this response. According to theories proposed by Ingber [ 83 ], the cytoskeleton as a whole reacts to changes in mechanical tension through the extracellular matrix, and the associated integrins, leading to the reorganisation of microfilaments and microtubules. In tandem with this, the cortical cytoskeleton, which supports the plasma membrane through the formation of a rigid 3D-framework, is in tension in an external mechanical field [ 84 ].
BioMed Research International
Therefore, by summarising the studies above Figure 1 , it is possible to see that practically all possible mechanisms of primary mechanotransduction depend on the condition of the submembrane cortical cytoskeleton structure which determines the mechanical properties of various types of cells, and this is ultimately reflected in stiffness of cells.
The changes in cell orientation within a gravitational field can be performed with the help of a horizontal clinostat and RPM—a random position machine [ 85 — 90 ].
- Wind in the Wires: A Woody Creek Novel 4;
- Mechanosensitivity in Cells and Tissues - NCBI Bookshelf.
- .
- ?
- Dein Vater er ist.: Systematik und Wirkung von Yodas Sprache in Krieg der Sterne (German Edition);
- Idylle à Santa Barbara - Le grand amour du Dr Freeman (Harlequin Blanche) (French Edition);
- .
Different types of cells have been cultivated under changing gravitational vector conditions. Most of the cells grown under these conditions showed changes in their cell profiles, as well as disorganisation of microtubules and microfilaments, and an increase in the number of apoptotic cells in the culture [ 91 — 96 ], in addition to alterations in mitochondrial localisation and clustering behaviour [ 56 ]. The data in the literature show that the influences of gravity vector changes in embryonic stem cells, decreased their ability to form embryoid bodies as a result of clinostation [ 89 , 97 ].
Conversely, according to data from the same authors, the clinostation of embryoid bodies significantly increased the number of beta-III tubulin-positive cells early neuroblasts. However, it also gave rise to an insignificant decrease in the number of MAP2-positive cells late neuroblasts over the course of spontaneous neuronal differentiation [ 89 , 97 ]. Mesenchymal stem cells are also sensitive to microgravity conditions; however, there is no common hypothesis in the interpretation of the results obtained from these studies [ 96 , 98 — ].
Modelling of microgravity effects, meanwhile, suggested an inhibition of osteogenic differentiation and activation of adipogenic differentiation of mesenchymal stem cells [ 88 , 96 , 98 — , — ]. The change in differentiation potential of stem cells, after real or modelled microgravity changes, can also be connected to cytoskeleton reorganisation, as some of cytoskeletal structures may take part in the determination of the differentiation pathway [ , ].
In addition, 24 hours of microgravity modelling caused transient changes in gene expression in mesenchymal stem cells. Some of these genes encoded actin cytoskeleton proteins and the elements associated with this. The modelling also decreased the capability for adhesion in these cells [ 90 ].
These changes are seen in alterations in division speed and development potential, and are probably connected with cytoskeleton reorganisation. Nevertheless, the primary levels of nonmuscle cellular responses to a change in external mechanical conditions are little studied and so the search for the mechanosensor will not end soon.
Results of numerous studies in conditions of real and simulated gravitational unloading testify that negative changes in various bodies and tissues are formed as a result of the action of microgravity. Skeletal muscles are especially vulnerable to a gravity-free state as the specialised organ that executes position and motor functions.
A subject of many studies is the m. In addition, a decrease in the functionality of both the whole muscle [ , ] and single fibres [ ] also takes place. Nevertheless, a substantive problem which is obstructing space development by humans, in particular flight to Mars, is the early readaptation period to gravity. Especially important is the reloading of muscle and cardiovascular system functionality, the acceleration of which is impossible without an understanding of the mechanism of development of adaptive changes.
Differently directed changes in the external mechanical conditions of skeletal and cardiac muscles of rats can be achieved by means of a common method; antiorthostatic hindlimb suspension of animals, according to the Ilyin-Novikov method with Morey-Holton modifications [ ].
On the one hand, the antiorthostatic suspension of animals leads to hindlimb disuse, while on the other hand, it causes an increase in the mechanical load on cardiomyocytes. In addition, the orientation of skeletal muscle fibres and cardiomyocytes in the gravitational field will change. According to the scarce data connected with the response of skeletal muscles cells to gravity disuse in the literature, one of the first events to occur, after two days of antiorthostatic disuse of the hindlimbs of mice, is the accumulation of calcium ions in the soleus muscle [ , ].
Above, we described that the maximal calcium ion accumulation in the soleus muscle fibres of rats and a Mongolian gerbils happens after one day of gravity disuse. For the medial gastrocnemius and tibialis anterior muscles, this maximum was seen a short time later—after seven days of antiorthostatic suspension [ ]. The increase in the resting calcium level can lead to calpain activation [ , ] and the following destruction of the muscle fibre structure.
However, the means by which it is accumulated remain unknown. Probably, this effect happens via L-type calcium channels, but the mechanosensitive channels of the TRP family may also take part. In any case, the functioning of the channels incorporated in the membrane depends on the condition of the sarcolemma and the cytoskeleton connected with it.
Moreover, we can suppose that the mechanical properties of cardiomyocytes will change, in other ways to that of skeletal muscle fibres, as the load on the cardiac muscle increases in microgravity conditions, while the load on the skeletal muscles of hindlimbs decreases. It is rather difficult to perform a direct evaluation of the native state of the cortical cytoskeleton of muscle fibres.
However, the definition of its mechanical properties transversal stiffness, to be exact can help in the analysis of its structural changes. In addition, the complicated sarcomere organisation of a muscle fibre enables us to suppose that the transversal stiffness of different parts of the sarcolemma Z-disk, M-line, and the part between them will differ from each other. The differentiation of stiffness factors is of great interest, for example, in regarding the signalling role of different proteins of the costamere, which probably depends on its structure. The methodology based on this above concept enabled us to define the transversal stiffness both of a contractile apparatus and of a membrane with a cortical cytoskeleton [ 5 ].
It was shown that the transversal stiffness of different parts of the soleus muscle contractile apparatus in the relaxed, calcium activated, or rigor states decreases, over the course of gravity disuse. It, in practical terms, does not change for the gastrocnemius muscle fibres and increases for tibialis anterior [ 8 , ]. Meanwhile, the transversal stiffness of the sarcolemma, with a cortical cytoskeleton in the relaxed state, decreases in all the muscles when under antiorthostatic disuse. Furthermore, for rat cardiomyocytes, it seems, that under the conditions of antiorthostatic disuse, there was an increase in mechanical load early on as a result of hypovolemia.
Consequently, the stiffness of the cortical cytoskeleton membrane increased: At reloading after antiorthostatic disuse, the stiffness of skeletal muscles fibres increased transversal stiffness of rat soleus muscle fibres: Changes in the transversal stiffness factors, both of skeletal muscles fibres and cardiomyocytes, under the conditions of antiorthostatic disuse and the following reloading, correlated with the content of nonmuscle isoforms of actin that form a cortical cytoskeleton in the membrane fraction.
Thus, acting on ion channels in the plasma membrane, mechanical stress can elicit a multitude of biochemical processes — both transient and long-lasting — inside a cell. This may ultimately influence the function of tissues and organs in health and disease. Several stretch-induced signaling cascades have been described with multiple levels of crosstalk between the different pathways. Increased sensitivity of the cells to mechanical stress is found under various pathological conditions. A detailed study of the underlying mechanisms may therefore help to identify novel therapeutic targets for a future clinical use.
Turn recording back on.
National Center for Biotechnology Information , U. Mechanosensitivity in Cells and Tissues Editors: For more information, see the Bookshelf Copyright Notice. Rosen, and Leonid V. Mario Pellegrino, Barbara Calabrese, M. Mechanosensitivity in Cells and Tissues. Similar articles in PubMed.